AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Which vsepr shapes are polar12/26/2023  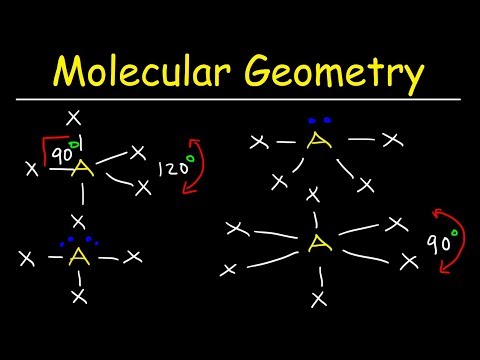 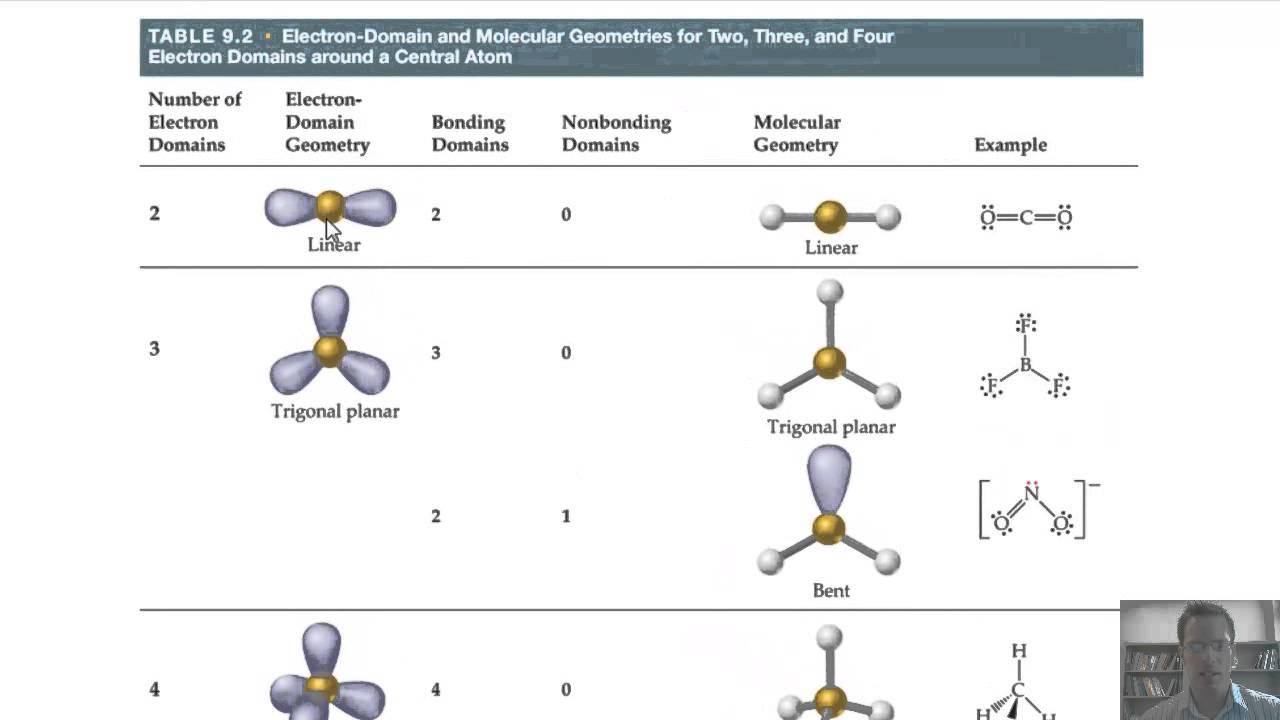
This is for the reason that when ch3cooh disintrigates acetate and H+ ion are produced. According to sp2 hybridization, the geometry of Ch3cooh will be planner.Ĭh3cooh is an ionic compound. In this compound central C uses sp2 hybrid orbital to make these bonds. O atom give their remaining unpaired electron to form C=O (pi) bonds. In the next step 1 O atom of –OH group, C atom of –Ch3 group and another O atom give their 1 unpaired electrons to form C-Ch3, C-OH and C-O bonds. When energy is applied, 1 2s electrons goes into vacant 2p orbital making a total of 4 unpaired electrons in the valance shell of C atom. In the valance shell of C atom we found that only 2 unpaired electrons present and to produce Ch3cooh, 4 unpaired electrons needed. 
Electronic configuration of C atom is 2s2 2p2 in ground energy state. Hybridization means where central atom mixes its atomic orbitals such as s, p, d which are in different energy state to produce similar energy hybrid orbitals e.g. there exist 6 valance electrons in O atom and H has only 1 valance electron.Ch3cooh contains total valance electron which will be equal to the summation of all the valance electron of C, O and H atoms. Hence in Ch3cooh (4*2)+(2*6)+(4*1)=24 valance electron present. Oxygen atom has the electronic configuration of 1s2 2s2 2p4 i.e. C has the electronic configuration of 1s2 2s2 2p2 and electronic configuration of C atom shows that there exist 4 electrons. To find out total valance electron in Ch3cooh it is must to know how many valance electrons present in C, H and oxygen atoms. These unshared electron pairs are shown in the lewis structure of Ch3cooh on C, H, O atoms as electron dots. Lone pair of electrons present on C atom of –C=O group= 4-4=0 i.e.no nonbonding electron pairs. Lone electron pair present on O atom of –OH group in Ch3cooh=6-2=4 i.e. Lone electron pair present on O atom of C=O group in acetic acid= 6-2=4 i.e. Lone electron pairs of an atom= electrons that present on the valance shell of the atom – how many are bonds formed by that atom. The formula with the help of which we can calculate the lone pair of electron on the given atom of Ch3cooh is depicted below: It is also known as nonbonding electron pair. The lone electron pairs are that valance electrons which are not shared between 2 atoms when they are connected through a covalent bond. From this we see that all the atoms satisfy octet rule expect H which satisfy doublet rule. The O atom of –OH group forms 2 σ bonds with C=O group and 1 H atom. The carbonyl O forms double bond with C atom and also contain 2 lone pairs. The carbonyl carbon form 1σ and 1∏ bond with O atom and also forms 2 σ bonds with -ch3 and –OH group. The C of ch3 group forms 3 bonds with H atom and 1 bond with C=O group. I n the Ch3cooh lewis structure we see that all the atoms fulfilled their octet. As the central atom in Ch3cooh is titled at 120 0 angle, the overall compound is planner.Ĭh3ccoh lewis structure angle Ch3cooh Lewis structure Octet Rule 120 0) and also O atom of –OH group is sp3 hybridized (i.e. The carbonyl O is same as that of carbonyl carbon which is sp2 hybridized (i.e. The carbon atom in –Ch3 group is sp3 hybridized i.e. In Ch3cooh lewis structure, carbonyl carbon uses sp2 hybrid orbital to form bonds with –ch3 group and –OH group i.e. Hence formal charge on central C atom is 0.Each atoms in Ch3cooh has 0 formal charge making the whole compound is electrically neutral. Hence formal charge on carbonyl C atom in Ch3cooh=4-8/2-0= 0.įormal charge on carbonyl O atom in Ch3cooh=6-4/2-4=0.įormal charge on O atom of –OH group=6-4/2-4=0.įormal charge on C atom of –ch3 group=4-8/2-0=0. The formal charge on the atoms in Ch3cooh can be obtained by the formula which is given below:įormal Charge (F.C) = No of valance electrons-(bonding electrons)/2- No of unshared pair of electron 
Ch3cooh exist in planner shape so that the bond angle found in acetic acid is 120 0. Ch3cooh lewis structure resonance Ch3cooh Lewis Structure ShapeĪccording to VSEPR theory the shape of Ch3cooh is trigonal planner in which central carbonyl carbon is surround by carbonyl O atom, -Ch3 group and -OH group.īut C of –Ch3 group is tetrahedrally surrounded by 3 H and –C=O group and also O atom of –OH group is tetrahedrally surrounded by –C=O group, H atom and 2 lone pairs. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
