AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 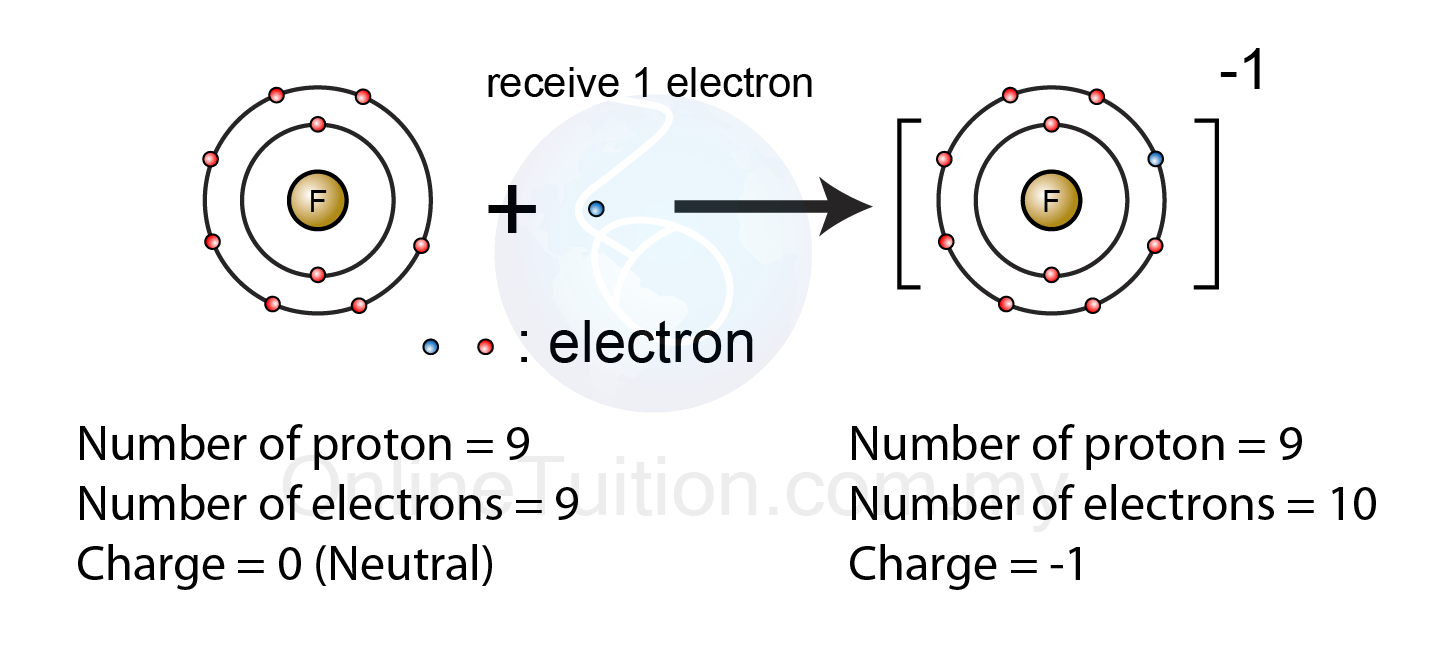
Because it has gained an electron, it has more electrons than protons so the overall charge of the chloride anion is negative. Similarly the Cl - ion is formed from the gain of one electron. We've always found it useful to think of the "t" in cation as a + symbol. Because it has lost an electron, it has more protons than electrons the overall charge of the sodium cation is positive. The Na + ion is formed from the loss of one electron. A sodium atom has 11 protons and 11 electrons. If an atom gains an electron, it also gains a negative charge (and becomes negatively charged) and is called an anion. If an atom loses an electron it also loses a negative charge (and becomes positively charged). They just have to gain or lose an electron. It's a fairly simple concept for an atom to become an ion. Salt, NaCl, contains positively and negatively charged atoms called ions. These atoms which are not neutral in charge are called ions. The compound sodium chloride has a positively charged sodium atom and a negatively charged chloride atom. An example we're all familiar with is good 'ole table salt, NaCl. In some cases, atoms can acquire an electrical charge. Just to be confusing, this is not a rule that is set in stone. The positive charge of the protons balances out the negative charge of the electrons and thus the overall net charge of the atom is zero. An atom is neutral, which means it contains the same number of protons and electrons. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
